Thermodynamics
Variables
Q= Energy transferred(Joules)
m= mass of material (kilograms)
c= specific heat capacity of material (Joules/ kilograms Celsius)
P= rate of energy transfer (watts)
Δt= Change in time(seconds)
k= thermal conductivity
A= area of thermal conductivity(meters sq.)
L= thickness of material(meters)
ΔT= Change in temperature(celsius)
Conduction Equations
Q=m*c*ΔT
Enrgey Transfer- The energy transfer eqaution is used to fiigure out how much energy(joules) is lost by the hotter object and gained by the colder object. Inversley you can also find unidentifed variables if you are given the Q such as mass.
P= kA*ΔT/L
P= Q/Δt
Rate of energy transfer- the rate of energy transfer equation gives you the average rate at which heat was being released or gained by an object. To get the rate all you need to know is the change in time and the total enrgy transfer. Once you divide the energy by the time(seconds) then you will get the average energy rate.
k=PL/A*ΔT
Thermal Conductivity- The thermal conductivity equation is used to find out how good of a energy conductor your material is. When you divede the Energy transfer rate by the area nad change in time you get the conductivity of the equation.
P/AΔT
U-value- The U value is the measure of the materials ability to conduct heat.
R= 1/U
R-value- The R value is the measure of the materials ability to ressist heat.
Q= Energy transferred(Joules)
m= mass of material (kilograms)
c= specific heat capacity of material (Joules/ kilograms Celsius)
P= rate of energy transfer (watts)
Δt= Change in time(seconds)
k= thermal conductivity
A= area of thermal conductivity(meters sq.)
L= thickness of material(meters)
ΔT= Change in temperature(celsius)
Conduction Equations
Q=m*c*ΔT
Enrgey Transfer- The energy transfer eqaution is used to fiigure out how much energy(joules) is lost by the hotter object and gained by the colder object. Inversley you can also find unidentifed variables if you are given the Q such as mass.
P= kA*ΔT/L
P= Q/Δt
Rate of energy transfer- the rate of energy transfer equation gives you the average rate at which heat was being released or gained by an object. To get the rate all you need to know is the change in time and the total enrgy transfer. Once you divide the energy by the time(seconds) then you will get the average energy rate.
k=PL/A*ΔT
Thermal Conductivity- The thermal conductivity equation is used to find out how good of a energy conductor your material is. When you divede the Energy transfer rate by the area nad change in time you get the conductivity of the equation.
P/AΔT
U-value- The U value is the measure of the materials ability to conduct heat.
R= 1/U
R-value- The R value is the measure of the materials ability to ressist heat.
Procedure
Team will desgin a renewable composite insulation material. The material will be put on top of a acrylic box with a light bulb inside of the box that will heat up and cool down the box based on how good your insulation is. You must record the rise of heat for 20 min. and the fall of heat for another 20 min and then determine certain values.
Materials
· Computer
· Heat box apparatus
· LoggerPro software
· 2 – Stainless steel temperature probes
· Insulation materials
· Tape
· Standard and metric ruler
· Heat box apparatus
· LoggerPro software
· 2 – Stainless steel temperature probes
· Insulation materials
· Tape
· Standard and metric ruler
Design
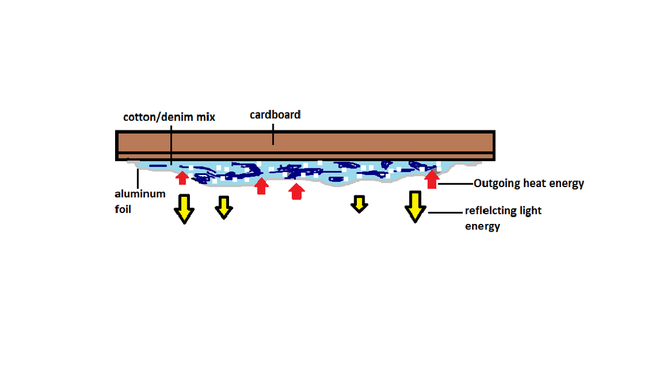
This is are insulation design that we made for this lab. It is composed of four diffrent materials. First we cut out a piece of cardboard that wa 22cm by 21cm that would be the base for the rest of the materials. After that we shredded some denim and cotton and made mixture that we laid down on the cardboard.this material was covered by a layer of aluminum that would be the side that would be facing the light bulb.
The point of our design is very simple but i think it is very efficent. The aluminum foil has two properties, It both conducts(absorbs) and reflects energy. The aluminum foil will reflect the light energy back into the box and this will probably make the box heat up alot. Once the 20min. of heating are up we will have 20 min. of cooling. During this time the aluminum foil will absorb most of the heat out and this is where the cotton and denim mix come in. They are ment to stop as much heat as they can and try to keep it in so it can stay warm as long as possible.The cardboard is a base for are materials but it will also help keep the outside temperature of the box the same.
Data
Inner dimensions:
Length: 14.4 cm = 0.144 m
Width: 14.3 cm = 0.143 m
Hight: 17.3 cm = 0.173 m
Volume = L*W*H = .144*.143*.173 = .003562416 m cu.
Length: 14.4 cm = 0.144 m
Width: 14.3 cm = 0.143 m
Hight: 17.3 cm = 0.173 m
Volume = L*W*H = .144*.143*.173 = .003562416 m cu.
Heat source (light bulb wattage): 25w
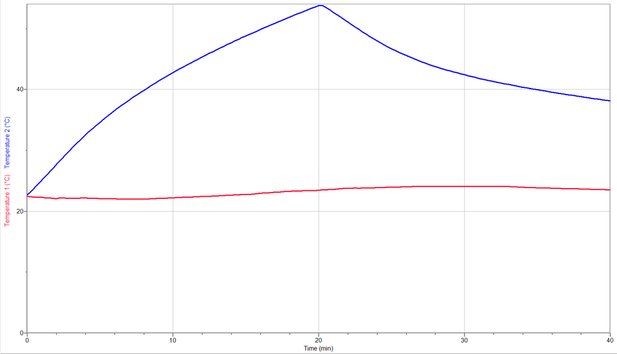
Initial internal temperature: 22.5 C
Maximum in ternal temperature: 55.8 C
Final internal temperature: 38.1 C
Inital room temperature: 22.7 C
Maximum room temperatue: 23.7 C
Final room temperature: 23.5 C
Heating time(s): 20 min
Cooling time(s): 20 min
Energy Gained by Air Energy Lost by Air Net Energy Retained
Q=m*c* change in T -Q= m*c*change in T Joules gained + Joules lost
Q= (.0036kg)(1000J/kg)(36.3C) -Q=(.0036kg)(1000J/kg)(17.7C) 130.68 J - 63.72 J
Q= 130.68 Joules Q= -63.72 Joules 66.96 Qnet
Initial internal temperature: 22.5 C
Maximum in ternal temperature: 55.8 C
Final internal temperature: 38.1 C
Inital room temperature: 22.7 C
Maximum room temperatue: 23.7 C
Final room temperature: 23.5 C
Heating time(s): 20 min
Cooling time(s): 20 min
Energy Gained by Air Energy Lost by Air Net Energy Retained
Q=m*c* change in T -Q= m*c*change in T Joules gained + Joules lost
Q= (.0036kg)(1000J/kg)(36.3C) -Q=(.0036kg)(1000J/kg)(17.7C) 130.68 J - 63.72 J
Q= 130.68 Joules Q= -63.72 Joules 66.96 Qnet
Conclusion
So overall are design worked very well and when compared to the other teams, we were among the higher Qnets. Are design really did work cause of our combination of both the aluminum foil and the denim/cotton mix. The aluminum foil has really good reflection of energy but it also losses energy fast we were able to negate the loss of most of the heat hence we were left with a really good insulation. this project really did teach me how thermodynamics works. I can see that people that make the insulation in the houses we live in really did take their time in creating it cause my house stays warm during the winter and cool during the summer. This is one way I see engineering start to apply to problems of daily life.